THESIS REPORT
Regulation of breast cancer cells’ bone-metastatic potential by mechanically stimulated osteocytes
Yu-Heng Vivian Ma
November 2019
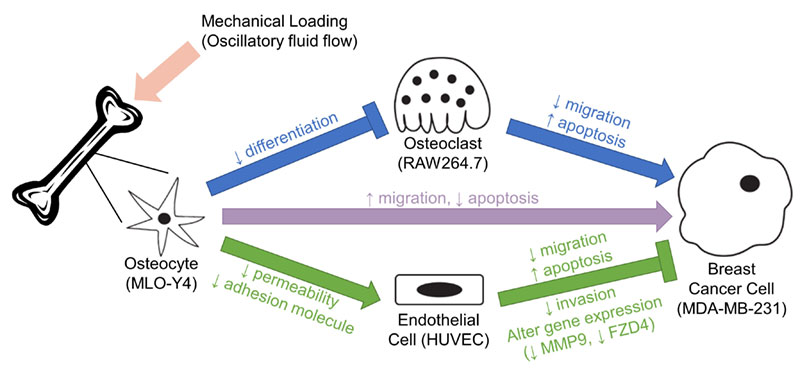
Bone metastasis, the migration of cancers to the bone, occurs in 65-75% of patients with advanced breast cancer and significantly increases patients’ morbidity and mortality. The bone-metastatic cancer cells interact with cells in the bone to disrupt the bone remodeling balance, causing reduced bone quality and other complications while facilitating tumor growth. Bone remodeling cells can be regulated by osteocytes, the major population of cells in the bone that are embedded in the bone matrix, in response to dynamic loading on the bone. Osteocytes also signal to blood vessel-lining endothelial cells that interact closely with cancer cells during early metastasis before a secondary tumor is established in the bone. Therefore, we hypothesized that mechanically stimulated osteocytes may regulate cancer cells directly and via other cells. To investigate, we mimicked what osteocytes experience in vivo during bone-loading activities, such as walking, with oscillatory fluid flow. We observed that factors secreted by flow-stimulated osteocytes increase cancer cell migration and survival. Contrastingly, signaling from flow-stimulated osteocytes through bone-resorbing osteoclasts or endothelial cells to cancer cells were anti-metastatic. Specifically, it reduced cancer cell migration, survival, and invasion. Factors secreted by flow-stimulated osteocytes also reduced cancer cells’ trans-endothelial migration and endothelial monolayers’ permeability and ability to be adhered by cancer cells. These demonstrated the capability of mechanically stimulated osteocytes in reducing the bone-metastatic potential of breast cancer cells by signaling through osteoclasts and endothelial cells. Investigating this regulation further can provide novel insights into the potential of bone-loading exercise in preventing bone metastasis.
Full text of Dr Y-H V. Ma’s thesis is available from HERE

Yu-Heng Vivian Ma
Institute of Biomaterials and Biomedical Engineering, University of Toronto

